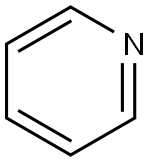
Pyridine
Content: ≥ 99%
Package: 200kg/barrel
Melting point: - 41.6 ℃
Boiling point: 115.3 ℃
Water solubility: soluble
Structural type:
Density: 0.983 g/cm ³
Uses: Pyridine can be used in industry as denaturant, dye aid, and raw materials for the synthesis of a series of products (including drugs, disinfectants, dyes, etc.).
Pyridine is a six membered heterocyclic compound containing one aza atom, that is, a compound formed by the substitution of one carbon in benzene molecule with nitrogen. It is similar to benzene in that it has the same electronic structure and still has aromaticity. Pyridine and its derivatives are more stable than benzene, and their reactivity is similar to nitrobenzene. Due to the electron absorption of nitrogen atoms in the ring, the electron density at positions 2, 4 and 6 is lower than that at positions 3 and 5. The typical aromatic electrophilic substitution reaction occurs at positions 3 and 5, but the reactivity is lower than that of benzene, so it is generally not easy to nitration, halogenation and sulfonation. In addition, these substitution reactions are carried out in acidic medium. Pyridine forms positively charged ions, which make electrophilic reagents inaccessible. The halogens of 2-or 4-halopyridines are active. Because of the low electron density at positions 2 and 6, nucleophilic substitution reactions can take place at these positions, such as reaction with sodium amino or potassium hydroxide, to give corresponding 2-aminopyridine or 2-hydroxypyridine. Pyridine can also be used as denaturant and dyeing aid in industry, as well as the starting material for synthesis of a series of products, including drugs, disinfectants, dyes, food seasonings, adhesives, explosives and so on. Pyridine is toxic, inhalation, ingestion or skin contact can lead to reduced male fertility, can also cause cancer.